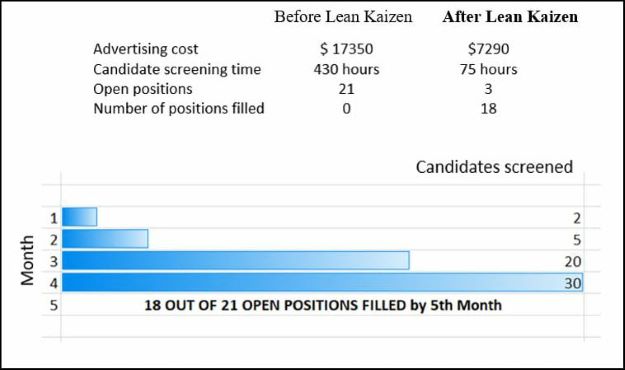
Many small organizations have human resources and/or budget constraints. But even with a small team of a single employee and an optimistic attitude of continual improvement, organizations can create operational excellence by installing Lean Kaizen.
Though, Lean Kaizen here has been illustrated in the pharma sector, the principles can be extended and used in any manufacturing sector.
Checkout the whitepaper...
Feel free to download this whitepaper for personal reading, but not for sale or resale. Hope you find it helpful. Reach out to me if you have questions.
Related Reading
- How to cut costs strategically using Kaizen
- Revolutionizing industries with Lean Six Sigma
- Streamline processes and workflows with Gemba Walk.
- Top Ten Strategic Decision-Making Tools for Operational Excellence
- Popular Continuous Improvement Workshops & Webinars for Manufacturing Industries
- Mistake-Proofing Pharmaceutical Product Development, Manufacturing and Logistics, Cost Savings via Poka Yoke
- Popular Continuous Improvement Tools.
Follow Shruti on Twitter, YouTube, LinkedIn
Categories: Kaizen | Life Sciences | Whitepaper
Keywords and Tags:
#Kaizenforpharma #LeanKaizen #StrategicKaizen #kaizen #operationalexcellence #continuousImprovement #ProcessImprovement #ProcessEfficiencyImprovement #BusinessProcessImprovement #BusinessTransformation #JapaneseKaizen #KaizenEvents #processoptimization